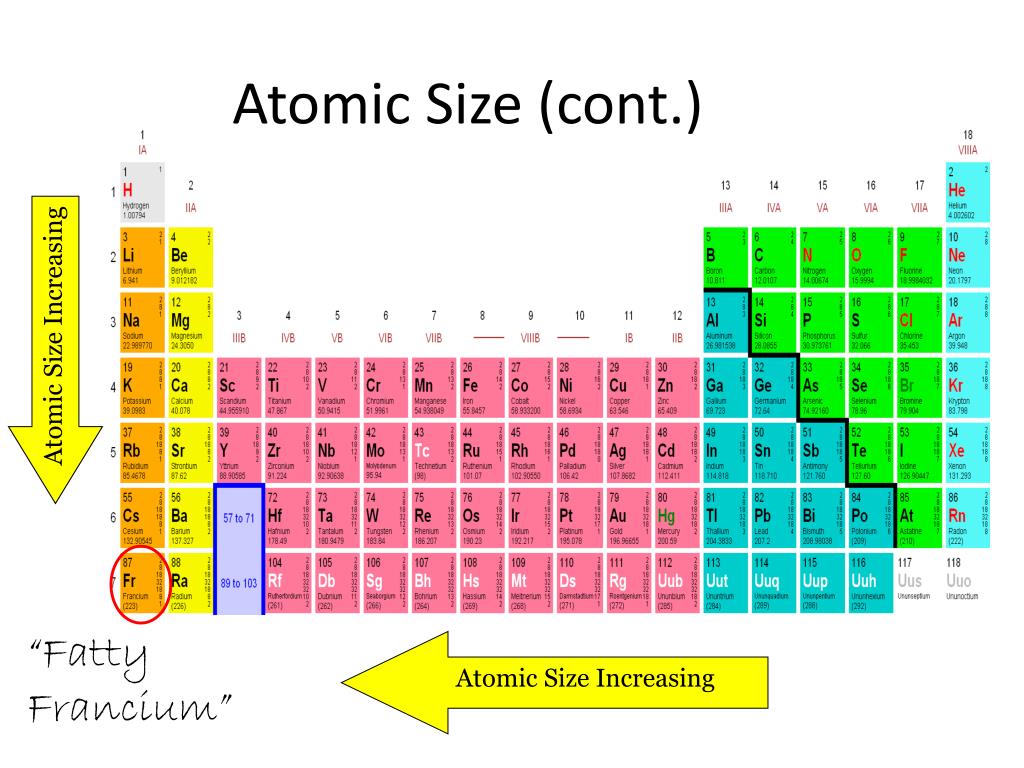
The effective charge (Zeff) of an atom is the net positive charge felt by valance. The following plot shows the variation of first ionization energies across the periodic table. periodic table, trends emerge that help explain how atomic radii changes. Consistent with size trends, first ionization energies generally increase across a period and decrease down a group. 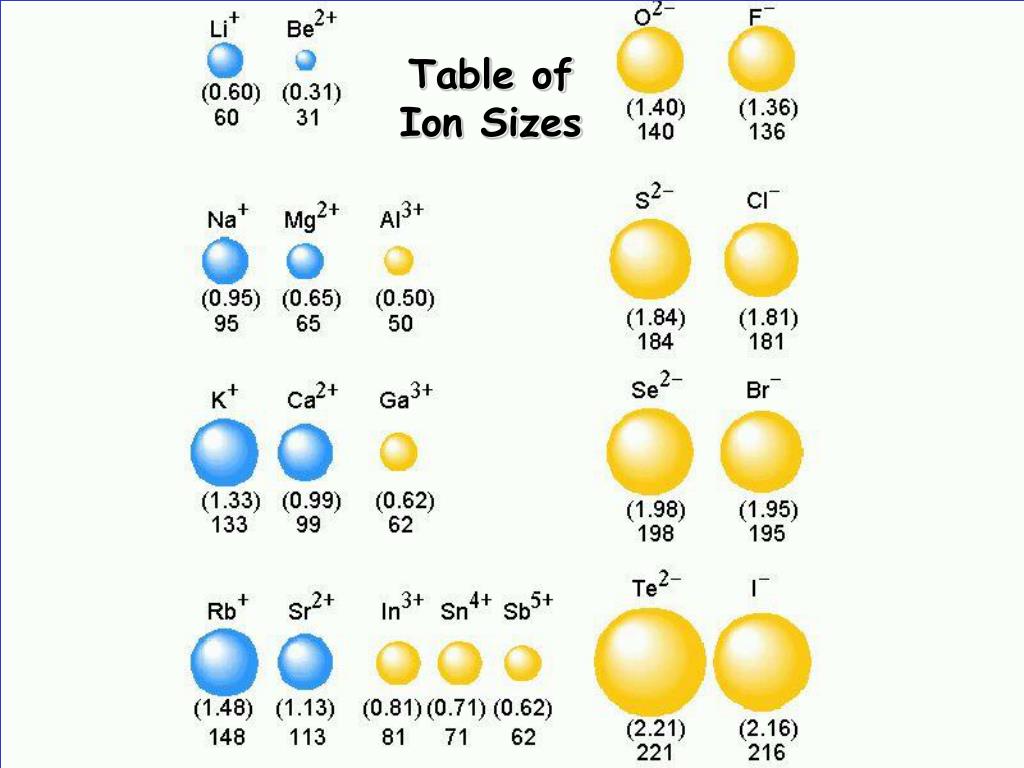
These data confirm the trends observed for metallic radii. Because of the way we organize the elements, there are special patter. The covalent radii of the main group elements are given in the figure below. The valence electrons are closer to the nucleus to which they are attracted in a smaller atom thus, more energy will be required to remove an electron by ionization. Why is the periodic table arranged the way it is There are specific reasons, you know. Trends in first ionization energies can be understood on the basis of size of atoms. \Īll ionization energies are positive, because it takes energy to remove an electron from the attraction of an atom’s nucleus. The first ionization energy, \(I_1\), refers to removing one electron from a neutral atom: Consequently, the size of the atom (and its covalent radius) must increase as we increase the distance of the outermost electrons from the nucleus. Ionization energy, I, is the energy required to remove an electron from a gaseous species. Increase in a shell causes increase in atomic size. When we move from from 3 d-series to 4 d-series, one extra shell is added in atom. Now, lets see the atomic size trend down the group. In the chart below, note the trends in the sizes (radii in picometers, pm) of the following ions with the same charge. So overall atomic size trend in the 3 d group looks like this. This similarity occurs because the members of a group have the same number and distribution of electrons in their valence shells. Ionic radii increase down a group for ions of the same charge. Describe and explain the observed trends in atomic size, ionization energy, and electron affinity of the elements The elements in groups (vertical columns) of the periodic table exhibit similar chemical behavior. This is an example of how these elements behave differently, and a reminder that trends do not necessarily apply to every instance.\): 69 pm Sections below cover the trends in atomic radius, first ionization energy, electronegativity, melting and boiling points, and density. Two elements decrease and then increase again, seemingly throwing the trend off. This page discusses the trends in some atomic and physical properties of the Group 1 elements - lithium, sodium, potassium, rubidium and cesium. Atomic size is difficult to measure because it has no definite boundary. Lesson Summary Atomic size is the distance from the nucleus to the valence shell. 
The atomic radius for the halogens increases down the group as n increases. Based on what you have learned, and without the assistance of the periodic trends. (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond. atomic radius: size of atom 2) Examine the trend in atomic radius from. On the bottom of the table we see a slight bump in the trend. Compared to the same graph for the elements in period 2, the graph for transition metals shows the trend for atomic radii is not as straightforward. The trends for the entire periodic table can be seen in Figure 12.1. However as we go down the group the situation of the equation is reversed and so our atoms become larger as their core charge gets smaller. This equation fits perfectly with the above diagram and explains why the atoms get smaller as they travel across the period. This is caused by the increase in the number of protons and. As this number gets higher, the valence electrons are pulled closer to the nucleus, therefore decreasing the atomic size of the atom. Atomic radius decreases from left to right within a period. The core charge is simply an expression of the attractive force that the centre of the nucleus gives off to the valence electrons. CORE CHARGE = PROTONS - NON-VALENCE ELECTRONS. Since 1899 the IUPAC Commission on Isotopic Abundances and Atomic Weights (CIAAW) has been evaluating atomic weights and abundances. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period.Ītomic radius can be linked to core charge. With the above image, courtesy of Webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.Ītomic radius is measured from the centre of the nucleus to the outermost electron shell. In general (but with a few exceptions) atomic size decreases from left to right and from the bottom to the top of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
